The Importance of Cleanroom Nitrile Gloves in Maintaining Contamination Control
In environments where a single microscopic particle can compromise millions of dollars in research or production, the choice of protective gear becomes critical. Cleanroom nitrile gloves serve as the first line of defence against human-borne contamination in these sensitive settings. Unlike standard gloves, cleanroom-specific variants are engineered to meet rigorous standards for particulate control, chemical resistance, and electrostatic discharge—making them indispensable in semiconductor manufacturing, pharmaceutical production, and biomedical research.
Understanding the Cleanroom Environment
Cleanrooms are controlled environments where airborne particles, temperature, humidity, and pressure are meticulously regulated. These spaces are classified under ISO 14644-1 standards, ranging from ISO Class 1 (strictest) to ISO Class 9. For instance, semiconductor fabrication often requires ISO Class 4-5 conditions, where ≤5,200 particles (≥0.5μm) per cubic meter are permitted. Human operators, however, naturally shed skin cells, oils, and microorganisms—introducing significant contamination risks. This is where specialised cleanroom gloves transition from accessory to necessity.
Why Nitrile? Material Advantages for Critical Applications
The debate between nitrile gloves vs latex heavily favours nitrile in cleanroom environments. While latex offers elasticity, it poses allergy risks and higher particulate shedding. Nitrile, a synthetic rubber, provides:
• Inherently Low Particulate Generation: Reduced fibre and particle release compared to latex or vinyl.
• Latex-Free Safety: Latex-free gloves eliminate protein-related allergic reactions.
• Superior Chemical Resistance: Withstands exposure to solvents, acids, and alkalis common in manufacturing and labs.
• Enhanced Durability: Higher puncture and tear resistance minimises failure risks during critical operations.
Key Specifications of True Cleanroom Gloves
Not all nitrile gloves qualify for cleanroom use. Authentic cleanroom nitrile gloves must meet stringent criteria:
Particulate Control
Gloves undergo testing per ISO 23464:2020 to measure total non-volatile residues (TNVR) and particle release. Premium gloves like INTCO Medical’s maintain TNVR <2.5μg/m² and ≤800 particles/cm² (for ≥0.5μm particles)—meeting ISO Class 4 standards.
Electrostatic Discharge (ESD) Safety
For electronics manufacturing, ESD-safe properties are non-negotiable. Gloves must demonstrate surface resistivity between 10^9-10^11 Ω/sq per ANSI/ESD S20.20 to prevent damaging sensitive components.
Material Purity
Advanced formulations avoid silicone, amino compounds, and plasticisers like dioctyl esters—substances that could outgas and contaminate processes.
Packaging Integrity
Sterile cleanroom gloves use double-barrier packaging (e.g., outer cardboard + inner vacuum seal) to preserve cleanliness until the point of use.
Consequences of Glove Failure in Controlled Environments
Using inadequate gloves risks:
• Product Contamination: Microbes or particles ruining batch integrity in pharmaceuticals.
• Process Failure: Static discharge destroying microchips during assembly.
• Regulatory Non-Compliance: Violating FDA cGMP or EU GMP guidelines, triggering audits or recalls.
• Financial Losses: Scrapped products, downtime, and cleanup costs exceeding glove investment.
INTCO Medical’s Cleanroom Glove Solutions: Engineered for Precision
As industries demand higher reliability, INTCO Medical—the world’s largest latex-free nitrile gloves manufacturer—leverages its expertise to produce cleanroom gloves that exceed industry benchmarks. Their disposable cleanroom nitrile gloves are manufactured in ISO-certified facilities with advanced cleaning processes to ensure:
Uncompromising Purity
With TNVR levels below 2.5μg/m² and ionic contamination controls, these gloves prevent introducing residues into sensitive environments.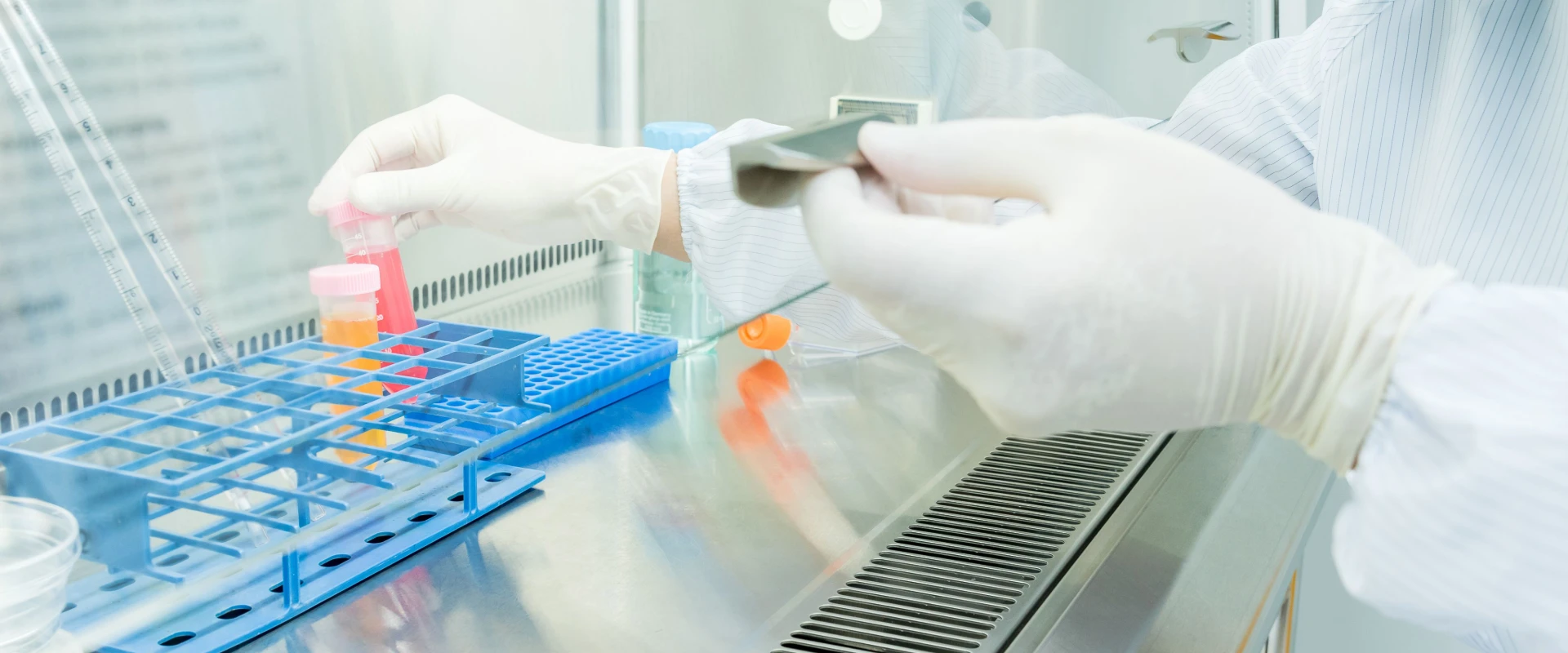
Dual Protection
Combining chemical resistance with ESD safety (surface resistance: 10^9-10^11 Ω/sq), they safeguard both processes and products.
Certified Performance
Compliant with ISO 23464:2020, EU RoHS, and ANSI/ESD S20.20, these gloves meet global standards for diverse applications.
User-Centered Design
Textured fingertips enhance grip for handling small components, while rolled cuffs simplify donning without tearing. The nitrile formulation ensures comfort during extended wear.
Applications Across Industries
• Semiconductor Manufacturing: Protecting wafers from particulates and ESD.
• Pharmaceutical Production: Maintaining sterility during drug formulation and packaging.
• Medical Device Assembly: Preventing biocompatibility issues from contaminants.
• Biomedical Research: Ensuring cell cultures and samples remain uncontaminated.
Partnering for Contamination Control
Selecting gloves isn’t merely a procurement decision—it’s a risk management strategy. INTCO Medical’s cleanroom gloves offer a validated solution for environments where failure is not an option. Their rigorous manufacturing and testing protocols assure that each pair meets the cleanliness and performance standards modern industries require.
Explore INTCO Medical’s Cleanroom Solutions
Discover gloves engineered for precision environments:
• Cleanroom Nitrile Gloves Overview
• Technical Specifications & Compliance
• Contact Our Cleanroom Specialists
FAQ
What are cleanroom nitrile gloves?
Cleanroom nitrile gloves are disposable gloves specifically designed for controlled environments. They are made from synthetic nitrile material and processed to minimize particles, residues, and contaminants, ensuring high cleanliness standards required in industries such as pharmaceuticals, electronics, and laboratories.
Why are nitrile gloves used in cleanroom environments?
Nitrile gloves are used in cleanrooms because they are latex-free, low in particulate generation, and resistant to chemicals. These properties help maintain contamination control while providing durability and protection for sensitive operations.
What contamination risks do cleanroom gloves help prevent?
Cleanroom gloves help prevent particulate contamination, microbial contamination, and chemical transfer. They act as a barrier between operators and critical environments, reducing the risk of product defects or compromised sterile conditions.
What standards define cleanroom nitrile gloves?
Cleanroom nitrile gloves are defined by compliance with standards such as ISO cleanroom classifications and ASTM testing requirements. High-quality manufacturers like INTCO Medical ensure gloves meet strict cleanliness, particle, and extractable limits.
How are cleanroom nitrile gloves different from regular nitrile gloves?
Cleanroom nitrile gloves undergo additional cleaning, processing, and packaging in controlled environments. Compared to regular nitrile gloves, they have lower particle counts, reduced ionic residues, and are often double-bagged for cleanroom entry.
How should cleanroom gloves be used to maintain contamination control?
Cleanroom gloves should be worn following strict gowning procedures, changed regularly, and handled carefully to avoid contamination. Proper donning techniques and controlled storage are essential to maintain their cleanliness level.
Are powder-free gloves required in cleanrooms?
Yes, powder-free gloves are required in cleanroom environments. Powder can introduce particles and compromise contamination control, so only powder-free, cleanroom-certified gloves are used.
What industries require cleanroom nitrile gloves?
Industries that require cleanroom nitrile gloves include pharmaceuticals, biotechnology, semiconductor manufacturing, medical device production, and precision electronics. These sectors depend on strict contamination control standards.
How does INTCO Medical ensure cleanroom glove quality?
INTCO Medical ensures cleanroom glove quality through advanced manufacturing processes, strict quality control systems, and compliance with international standards. Their cleanroom gloves are produced and packaged to meet the requirements of controlled environments worldwide.