OEM vs Branded Gloves: 2026 Guide for Distributors, Importers, and Private Label Buyers
Choose Branded when your accounts are compliance-heavy (healthcare, tenders) and you want fewer documentation “moving parts.”
Choose OEM / Private label when you need margin and brand control—and you can run a repeatable documentation + labelling + supplier qualification process.
Choose Hybrid when you want branded coverage for high-liability channels and OEM pricing power for cost-sensitive channels.
If you distribute or import disposable gloves in 2026, “OEM vs branded” isn’t a quality debate—it’s a business and compliance decision.
The same high-capability factories can make both OEM (private label) and branded gloves. The difference is who owns the regulatory file, the labelling claims, and the risk when something goes wrong.
This guide is written for distributors, importers, wholesalers, and private label buyers who need to balance margin, speed to market, documentation, and liability—especially in regulated verticals.
2026 context: why this decision got sharper (FDA QMSR 2026, EU MDR, and AQL)
Three forces are raising the bar:
-
Stricter quality-system expectations in the U.S. The FDA’s Quality Management System Regulation (QMSR) became effective on February 2, 2026, incorporating ISO 13485:2016 into 21 CFR Part 820 (with FDA-specific clarifications). See the FDA’s overview of the Quality Management System Regulation (QMSR): FDA QMSR.
-
Ongoing EU MDR enforcement pressure. Under EU MDR, importers and distributors have defined obligations as economic operators. The European Commission summarises these responsibilities here: EU MDR economic operators.
-
Higher expectations for consistency and traceability. Buyers are asking for tighter AQL targets, clearer lot traceability, and faster “show me the docs” turnaround.
What are OEM vs branded gloves?
OEM (private label) gloves
OEM (Original Equipment Manufacturer) / private label means a manufacturer produces the gloves, but the distributor/private label buyer owns the brand and packaging (and often the compliance positioning in-market).
In practice, OEM often includes custom packaging, labelling, and product spec selection (material, thickness, texture, cuff style), plus ongoing lot documentation.
If you’re building a private label program, you’ll typically start with a manufacturer’s standard line and then customise packaging and (sometimes) specs. INTCO Medical, for example, is the largest disposable glove manufacturer in the world and publishes an overview of its private-label capabilities under disposable gloves OEM/ODM services.
Branded gloves
Branded gloves are sold under an established brand’s name. The brand typically controls product claims, labelling, and the documentation package—while manufacturing may still be done by large OEM factories.
For a distributor, branded programs often mean faster onboarding, smaller MOQs, and less work in compliance operations (because the brand has already built the structure).
The structural difference (the part buyers miss)
Many glove sourcing problems come from confusing manufacturing capability with ownership of the regulatory/commercial system.
-
The manufacturer makes the product and runs quality control.
-
Brand owner: owns labelling claims, spec positioning, and typically the compliance file for that branded SKU.
-
Distributor/importer/private label buyer: sells into a market and may become the party that must prove the product meets local rules.
OEM vs branded gloves: quick answer
OEM is usually best when
-
You need margin control and pricing flexibility.
-
You want private label positioning (retail, e-commerce, or regional distribution).
-
You can operate (or outsource) documentation control, labelling review, and supplier qualification.
Branded is usually best when
-
Risk reduction matters more than margin.
-
You need a fast go-to-market.
-
You want more of the compliance burden to sit with the brand owner (especially in medical distribution).
A hybrid strategy often wins in 2026
A hybrid is common when you want:
-
branded SKUs for high-liability accounts (healthcare, regulated tenders)
-
OEM/private label SKUs for price-sensitive channels (food service, industrial, retail)
Head-to-head comparison (the criteria that actually move the needle)
|
Evaluation criteria |
OEM / Private label |
Branded |
|---|---|---|
|
Margin potential |
Higher upside |
Typically lower |
|
MOQ / setup effort |
Higher MOQs + packaging setup |
Lower MOQs; faster onboarding |
|
Compliance workload |
Usually higher for the importer/distributor |
Usually lower for the distributor |
|
Documentation speed |
Depends on your supplier + your internal process |
Often standardised by the brand |
|
Product consistency |
Determined by factory QC and process control |
Also determined by factory QC and process control |
|
Market trust |
You must earn it |
The brand carries it |
Key takeaway: Branding doesn’t create consistency—manufacturing systems do. Your model choice decides who is responsible when consistency or documentation breaks.
Cost and margin
OEM: more control, more responsibility
OEM/private label can deliver a higher margin because you own:
-
pricing strategy
-
packaging and positioning
-
SKU assortment (good/better/best)
But you’ll usually pay for that control in:
-
higher MOQs
-
packaging/tooling lead time
-
added QA and compliance operations
Branded: less upside, fewer surprises
Branded gloves can be easier to run operationally:
-
predictable price lists
-
established packaging and claims
-
less internal time spent building documentation packets
You give up some margin, but you may gain speed and reduce the chance of “hidden work.”
AQL and performance consistency
AQL (Acceptable Quality Level) is a sampling-based approach used in lot acceptance to define the maximum acceptable defect rate for a given sampling plan. In glove QC, glove AQL is most commonly discussed in the context of barrier integrity (e.g., pinhole/leak defects) and lot release decisions.
To avoid “AQL as marketing,” align AQL targets with the performance standards you are buying to. Common glove standards used in procurement and compliance discussions include:
-
U.S. (ASTM) medical glove specifications (material-specific), such as ASTM D6319 (nitrile exam gloves), ASTM D3578 (rubber/latex exam gloves), ASTM D5250 (vinyl exam gloves), and ASTM D3577 (rubber surgical gloves).
-
Chemotherapy drug permeation resistance is commonly evaluated using ASTM D6978 when gloves are marketed for chemo handling.
-
EU medical gloves for single use are commonly aligned to EN 455 (Parts 1–4).
OEM vs branded: what actually changes
-
OEM: Your consistency is as good as your manufacturer’s process control and your ability to enforce spec discipline across orders.
-
Branded: Consistency tends to be more stable because the brand limits variation (fewer spec changes, standardised QC expectations).
But the manufacturing reality is simple:
Key takeaway: The factory’s quality system is the primary driver of performance. A branded box does not automatically mean a better production line.
Documentation and claims integrity
This is where OEM vs branded diverges the most.
OEM: You own the claim stack
With private label gloves, you (or your legal entity in-market) may be responsible for ensuring:
-
labelling claims match test evidence
-
Your packaging meets local requirements
-
You can produce documentation during audits or customer qualification
In medical markets, the FDA notes that medical gloves are regulated as Class I reserved medical devices that require a 510(k). See: FDA medical gloves overview.
Practical takeaway for OEM programs: build a process for verifying clearance and documents—don’t rely on verbal assurances.
Branded: the brand typically owns the documentation package
For branded gloves, distributors usually receive a standardised documentation set. That doesn’t eliminate your responsibilities, but it reduces the number of “moving parts” you must maintain.
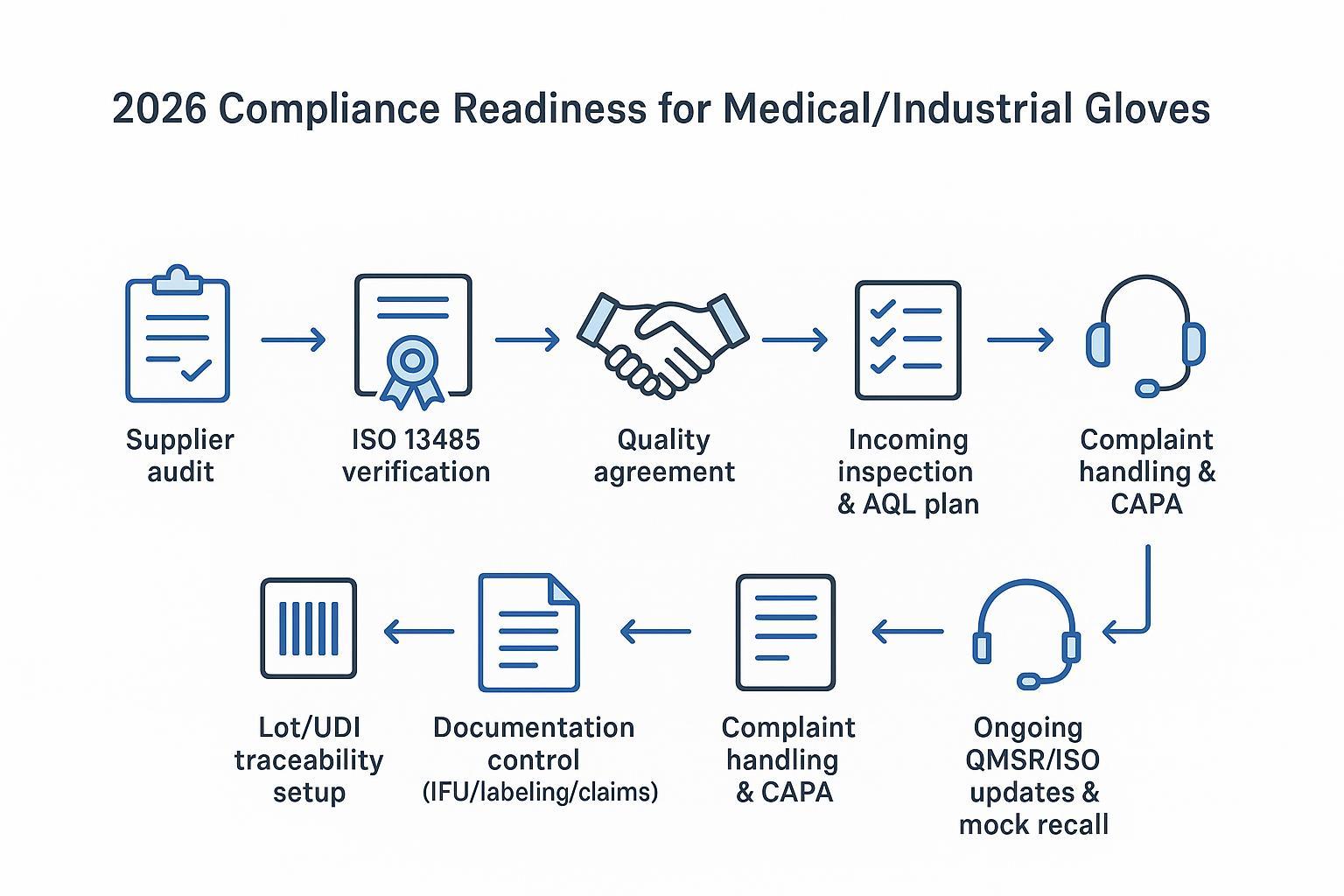
Compliance, liability, and QMSR readiness in 2026
510(k) and verification workflow (U.S. medical exam gloves)
If you distribute medical exam gloves in the U.S., the safest operational posture is to treat 510(k) verification as a repeatable checklist:
-
Obtain the 510(k) number for the exact SKU.
-
Verify it in the FDA’s 510(k) database: FDA 510(k) Premarket Notification search.
-
Match the device description (material, intended use) to your spec and labelling.
EU MDR: importer/distributor obligations don’t disappear
If you place a product on the EU market, MDR assigns defined obligations to economic operators. In MDR (Regulation (EU) 2017/745), Article 13 outlines general obligations of importers, and Article 14 outlines general obligations of distributors. A useful starting point is the European Commission’s overview of economic operators (linked above).
QMSR: what it changes (in practical terms)
QMSR is not just “new acronyms.” It raises the cost of weak systems.
Even if you’re not a manufacturer, your private label program will be judged by how quickly you can produce:
-
controlled documentation
-
lot of traceability evidence
-
complaint handling records
-
supplier qualification artifacts
The FDA’s QMSR overview is the canonical reference (linked above in the 2026 context section).
QMSR readiness checklist (OEM programs)
Use this checklist when qualifying a factory for medical or high-risk industrial accounts:
-
ISO 13485-certified QMS (when applicable to your market/category)
-
defined incoming inspection + lot release process (including AQL expectations)
-
full lot traceability (you can map a shipped case back to production lot)
-
documented complaint handling and CAPA path
-
recall readiness (mock recall capability, not just “we can recall”)
Supply, MOQs, and go-to-market
MOQs and lead times
-
OEM: typically higher MOQs, longer lead times (packaging approvals, artwork, and setup).
-
Branded: lower initial thresholds and faster replenishment cycles.
If you’re dual-sourcing, align MOQs to channel risk:
-
reserve OEM programs for stable, forecastable demand
-
Use branded suppliers to backstop volatility and urgent orders
Supply risk mitigation (works in both models)
-
qualify at least two factories (or factory + brand) for core SKUs
-
diversify production geography where feasible
-
Maintain safety stock for high-velocity sizes
Which model should you choose (by use case)
Healthcare and medical distribution
-
Recommended: Branded or Hybrid
-
Why: strict compliance expectations and higher liability sensitivity
Food service and retail
-
Recommended: OEM or Hybrid
-
Why: cost sensitivity with moderate compliance needs
Industrial and manufacturing
-
Recommended: OEM
-
Why: high volume demand and specification flexibility
E-commerce and private label brands
-
Recommended: OEM
-
Why: brand control and margin optimisation
Where INTCO Medical fits
When distributors need an OEM/private label pathway, the most important screening question is not “OEM or branded?” but “does the manufacturer operate at the documentation and traceability level your customers require?”
INTCO Medical positions itself as a large-scale glove manufacturer with OEM/ODM programs (see the OEM/ODM services page linked above) and publishes product/category pages (e.g., disposable nitrile exam gloves) that can be used as starting points for spec alignment and documentation discussions.
Conclusion: OEM vs branded is a strategy choice, not a quality choice
-
OEM vs branded is mainly about control vs responsibility.
-
Consistency comes from manufacturing systems, not the logo on the box.
-
Hybrid sourcing is becoming the default in 2026 because it balances margin, speed, and supply resilience.
Next steps
-
Map your verticals by compliance risk and decide which SKUs must be branded vs OEM.
-
Build a documentation checklist (510(k), test reports, traceability, complaint/CAPA process).
-
Qualify at least two supply paths for top-selling sizes and materials.
For deeper supplier qualification criteria, you can also reference INTCO’s related checklist-style article: How to Choose a Medical Glove Supplier: Buyer’s Guide 2026.
FAQs
Are OEM gloves safe for medical use?
Yes—if the exact glove SKU meets applicable requirements (e.g., performance standards, documentation, and the correct regulatory pathway for the market). In the U.S., medical gloves require a 510(k); the FDA medical gloves overview linked above is a good starting point.
OEM vs branded gloves: which is more cost-effective in 2026?
OEM is usually more cost-effective at scale because you control the cost stack and pricing. Branded is often more cost-effective operationally when you factor in speed and reduced compliance workload.
Do OEM gloves meet FDA 510(k) requirements?
They can, but you should verify the clearance for the exact SKU in the FDA’s database (the 510(k) search tool linked above).
Who is liable for defects in private-label gloves?
It depends on jurisdiction and contractual structure, but OEM/private label programs commonly place more responsibility on the importer/private label brand owner. Treat liability allocation as a legal question and confirm it in your contracts.
Can distributors switch from branded to OEM sourcing?
Yes. The operational work is mainly in supplier qualification, packaging/labelling controls, documentation readiness, and a repeatable audit/traceability process.
What is the MOQ difference between OEM and branded gloves?
OEM typically requires higher MOQs and longer lead times due to packaging customisation and setup. Branded programs often allow smaller initial buys and faster replenishment.